
The trial must have gone to plan as the company is making the SaaS ELN, known as Collaborative Electronic Research Framework (CERF), available immediately, providing researchers with a means to securely share information across the web, as well as secure record keeping.
CERF And Security
Rescentris is being coy about who developed their SaaS product, referring to them only as “SaaS experts”, but maybe that’s just part of the whole security thing. We say that because secure is one of its key features including a HIPAA and SAS 70 compliant data center with dedicated enterprise CERF server instances and databases.
Why all the security? The people who are expected to avail of the service are those at the cutting edge of R&D of some of the biggest companies in the world, who tend to get hysterical when their Intellectual Property risks being compromised.
However, that’s only a very hypothetical threat with ELN, which provides records and document management, role-based access controls, time/date stamping, audit trails, digital signatures, witnessing and PDF reporting for patents and eDiscovery. All that for US$ 50 per licence per month, including e-records archiving.
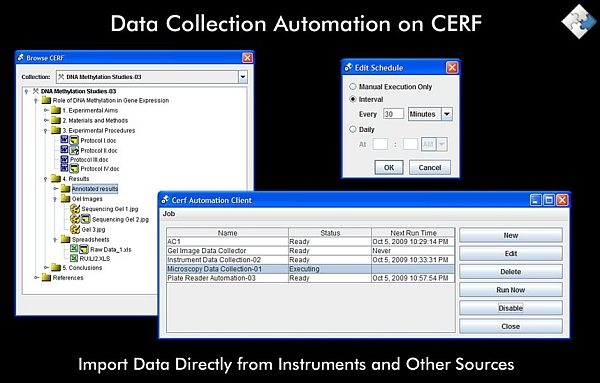
Secure Knowledge Management
Let’s take a look under the hood. CERF is an Electronic Lab Notebook that uses knowledge management technologies to enable researchers across global enterprises to collaborate -- even in the short term -- in a secure environment.
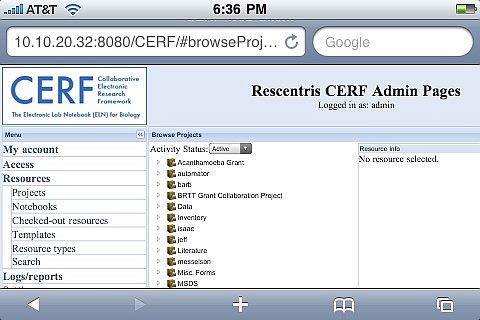
CERF: Secure administration
The SaaS version gives them cloud-based software enabling the exchange of data and documents, in all their versions across entire enterprises without risking possible security issues with email.
Operating through a client that can be installed on both Windows and Mac and accessed inside or outside of an organization depending on permissions, once users have logged in they can add any kind of data which is moved across to the CERF service and indexed on request according to 21CFR11 regulations.
CERF: Remote client access
Just to recap on 21CFR11, they are the regulations that provide criteria for acceptance by the FDA of electronic records, electronic signatures and handwritten signatures executed to electronic records -- in other words they become to all intents and purposes, records.
Features that come with CERF include:
- Data capture using templates, ontologies, controlled vocabularies and metadata for categorization and archiving.
- Search abilities using semantic technologies across unstructured and structured data to maximize access to data.
- Record creation for patent reviews, and for dissemination of discoveries across wider audiences.
- Easy access to knowledge and intellectual property with an open framework that allows for uses and development with future technologies.
The bottom line is that CERF gives research based organizations easy methods of not only marketing new discoveries, but also methods of protecting that information with automatic record creation by registering content, metadata and activities, as well as time/date stamping and creating audit trails.
